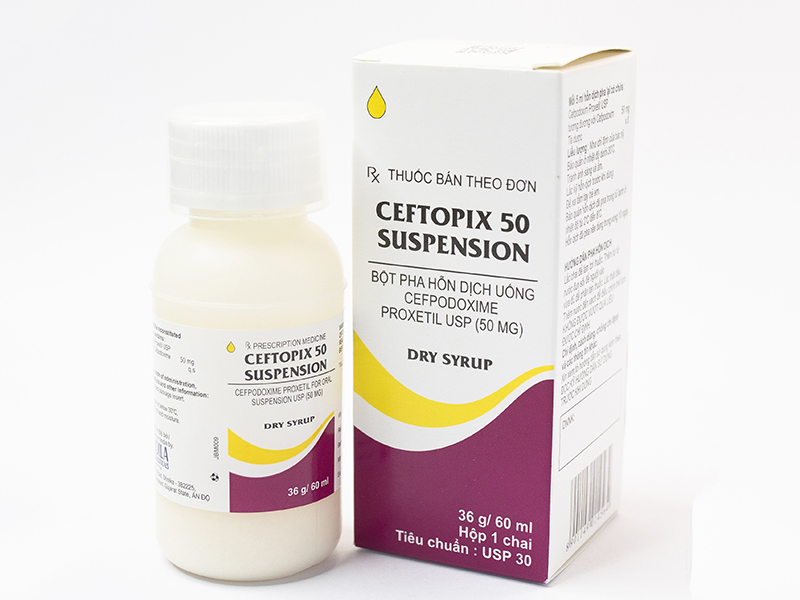
- Drug name: Ceftopix 50 Suspension
- Active ingredient name, Concentration – Content: Cefpodoxime 50 mg/5ml
- Specification, Dosage form, Route of administration: Box of 1 bottle of 36g, powder for oral suspension
- Expiry date (month): 24 months
- Registration number or license: VN-17290-13
- Manufacturing facility – manufacturing country: CADILA Pharmaceuticals Limited- India


Gửi bình luận